Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
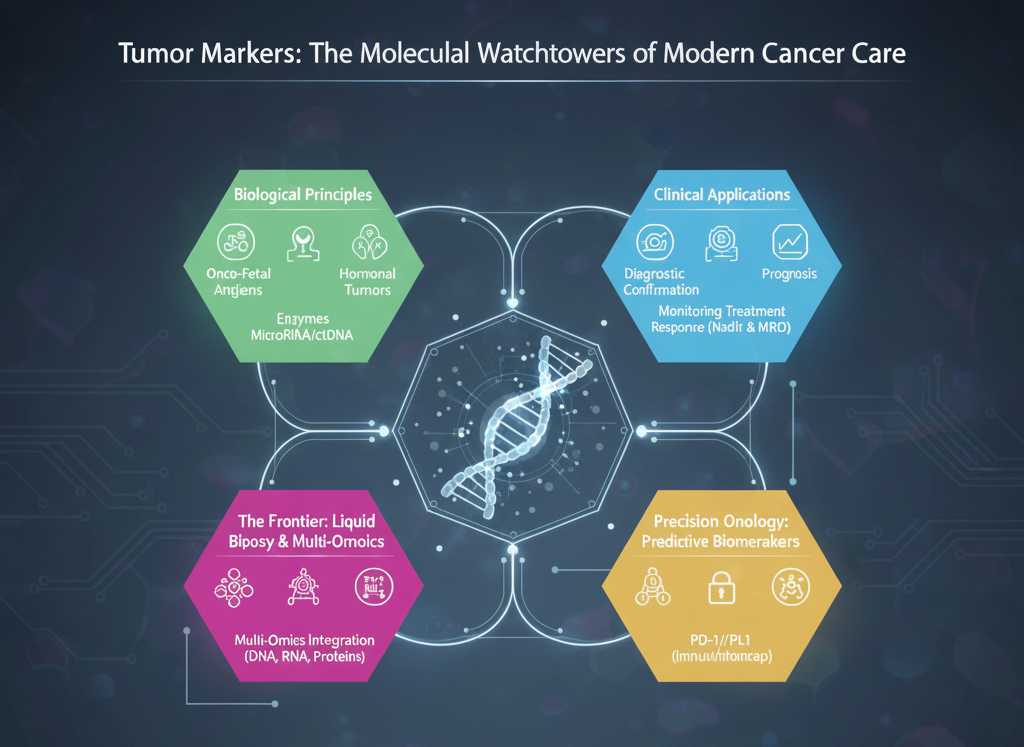
In the past, cancer medicine depended upon a generic and late-stage indicator; today, the use of tumor markers encompasses a sophisticated technique of “precision medicine,” from the time of diagnosis to post-therapy follow-ups.
Indeed, a tumor marker is not a “species” of a molecule. It is a “concept,” a means of identifying the kind of cancer through various molecular expressions.
These are proteins normally produced during fetal life but absent in the adult. When cancer occurs, the cell undergoes a “reversion” to produce these proteins again. Examples include Alpha-fetoprotein (AFP) for liver cancer and Carcinoembryonic Antigen (CEA) for colon cancer.
Some tumors, especially those of the endocrine system, produce excessive quantities of a hormone. The presence of high calcitonin, for instance, is a very specific indicator of the malignancy of the epithelial lining of the thyroid.
Some malignancies produce excess quantities of specific enzymes; these are secreted into the bloodstream. The most notable of these is the use of the Prostate-Specific Antigen (PSA) for prostate health.
As a spin-off of modern molecular biology, circulating tumor DNA (ctDNA) and MicroRNA (miRNA) have been established as highly sensitive biological markers employable by the technique of “liquid biopsy.”
Biological markers used in cancer diagnostics must be understood in the clinical context of diagnosing specific conditions. Unlike other imaging diagnostics, biological markers give a “dynamic view” of cancer.
While not specific for the general population, tumor markers are indispensable tools for screening high-risk families, such as the timing of HCC (Hepatocellular Carcinoma) surveillance in chronic liver disease patients.
Biological markers give a “biological clue” for a histopathometric study of a tissue biopsy specimen. For instance, in a patient presenting with a pelvic mass, a high CA-125 level is specific to an ovary vs. a gastrointestinal primary.
The level of a tumor marker at the time of diagnosis is often reflective of the “tumor burden” or aggressiveness.
4. Monitoring Treatment Response
This is perhaps the most common use of a tumor marker in current clinical practice. If a treatment schedule is effective, a decrease in the tumor markers should be observed.
“A Nadir”: Following surgery and chemotherapy, the level of the tumor marker should decrease to its “nadir.” However, if it persists, it signifies Minimal Residual Disease (MRD), which reflects a surviving “cloud of cancer cells.”
The year 2025 marks a new milestone, with a transition from a single protein test to a “liquid biopsy.”
The Benefit of a “Liquid Biopsy”: No longer will a “needle biopsy” of a tumor in the lungs or liver be necessary. Today, we are able to sequence ctDNA in a blood test.
Real-time Monitoring: It allows us to monitor clonal evolution in real time. Cancer cells will mutate to develop resistance to a drug, and these markers catch that shift early.
Multi-omics Integration: The ability to integrate DNA sequencing, Transcriptomics (RNA), and Proteomics is facilitating the development of “signature panels.”
This creates a new “digital profile of cancer,” essentially a digital biopsy that analyzes dozens of cancer biomarkers at once to solve the cancer puzzle.
HER-2/neu: These markers are used to assess the responsiveness to specific drugs in Breast Cancer and Gastric Cancer. A patient is likely to respond to targeted drugs such as Trastuzumab.
PD-1/PD-L1: We are at a critical moment in cancer treatment where this marker is vital for Immunotherapy. It acts as a predictor of a treatment response, signaling an “immune system unleashing treatment.”
There are no perfect tumor markers. There are also two major biological challenges in this area:
Biological Challenge – Sensitivity: Some early-stage tumors may not produce sufficient amounts of their associated markers to be measured.
Biological Challenge – Specificity: Most markers can be present in non-cancerous situations as well. For instance, PSA can be present when the prostate is enlarged because of inflammation (Prostatitis), resulting in “false positives” and panic.
The Information Challenge: As we begin to create “Pan-Cancer” screening tests capable of detecting signatures of several types of cancer from a single blood sample, there arise profound consequences in dealing with “over-diagnosis“—detecting early-stage tumors that may never have caused the patient any harm in his lifetime.
| Feature | Traditional Tumor Markers (CEA/PSA) | Next Generation Tumor Markers (ctDNA/Exosomes) |
| Structure | Mostly Proteins in Nature | Nucleic Acids or Vesicles |
| Invasiveness | Blood/Urine (Non-Invasive) | Blood/Saliva (Non-Invasive) |
| Sensitivity | Moderate (mostly late-stage) | Higher (Early Mutational identification) |
| Dynamic Range | Reflects Tumor Burden quantity | Reflects underlying mutational targeting |
| Role in Resistance | Unknown | Identifies resistant mutations (e.g., KRAS) |
Today, tumor markers go beyond their traditional use as catchwords in medical literature to being action facilitators in these decision-making moments in the art of cancer medicine. By now utilizing the awesome power of biotechnology and Artificial Intelligence, we are turning these molecular watchtowers into the definitive map for a cure.